Abstract
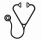
Introduction
Blast-phase (BP), or leukemic transformation is a rare and devastating complication of myeloproliferative neoplasms (MPNs) (primary myelofibrosis (PMF), polycythemia vera (PV), essential thrombocythemia (ET), and post-PV/ET myelofibrosis). Patients with BP-MPNs have a poor prognosis with a median overall survival of less than 6 months, and there is no standard treatment regimen for these aggressive diseases (Dunbar et al, Blood. 2020). The development of hypomethylating agent (HMA)/venetoclax (Ven) combination offers new hope for some with AML, but has been relatively disappointing in BP-MPN, though data are limited and retrospective (Masarova et al, Blood Adv. 2021; Gangat et al, Am J Hematol. 2021). Here, we add our experience with several common treatment regimens for BP-MPN.
Methods
We retrospectively analyzed data from 39 consecutive patients with BP-MPNs diagnosed from December 2008 to February 2021 who received treatment at the University of Michigan. We included all patients with a previous diagnosis of MPNs who had ≥ 20% blasts in the peripheral blood or bone marrow, and subsequently received systemic therapy. One patient with a myeloid sarcoma was included as well. Disease characteristics at time of BP-transformation were noted. Patients were divided into the following groups based on 1 st-line induction therapy: 7+3 (daunorubicin and cytarabine), FLAG (fludarabine, high-dose cytarabine and G-CSF), hypomethylating agent only (decitabine or azacitidine), and HMA/Ven. Patients were followed for 2 years post-diagnosis. Differences in induction response were assessed using the Chi-square test. Differences in overall survival were calculated using the Kaplan-Meier regression with the log-rank test. Two patients who received alternate induction therapy outside of the four groups were not included in these analyses.
Results
The composite BP-MPN population had a median advanced age of 69 years old and a median ECOG performance status (PS) of 1. Most (97.4%) had received systemic treatment prior to their transformation for their MPN, with 71.8% receiving hydroxyurea and 41.0% receiving ruxolitinib. The rate of response (CR, CRi, MLFS) was highest in the HMA/Ven group at 42.9%, followed by FLAG (29.4%), HMA only (11.1%), and 7+3 (0%), p = 0.033 (Table 3). Despite the higher response rate, differences in 2-year OS were not significantly different among the 4 groups: 7+3 (25.0%), FLAG (7.7%), HMA (0%), HMA/Ven (20.0%) (p=0.92, Figure 1). Median time to relapse after achieving remission ranged from 2-10 months, and did not vary significantly based on induction regimen. Patients in the HMA and HMA/Ven groups had higher incidences of death with induction at 77.8% and 28.6%, respectively (Table 3).
Conclusions
The highest rates of response, including complete remission, were achieved with the combination of HMA and venetoclax compared to intensive induction chemotherapy or HMA alone. However, this did not translate into significant differences in OS, which is consistent with other retrospective reports. No responses were seen with 7+3 induction, though several patients were able to go on salvaged with other therapies and subsequent allogeneic stem cell transplantation thereafter. Finally, the baseline poor ECOG PS of the HMA group and borderline ECOG PS of the HMA/Ven group also contribute to their low survival rates.Larger, prospective studies comparing currently available treatment regimens in BP-MPN would be helpful, but ultimately new therapies are desperately needed for this high-risk disease.
Bixby: Takeda: Consultancy. Talpaz: Constellation: Membership on an entity's Board of Directors or advisory committees; Bristol Myers Squibb: Membership on an entity's Board of Directors or advisory committees; Takeda: Other: Grant/research support ; Celgene: Consultancy; Imago: Consultancy; Novartis: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding.
Author notes
 This icon denotes a clinically relevant abstract
This icon denotes a clinically relevant abstract

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal